The Hallmarks of Copper Single Atom Catalysts in Direct Alcohol Fuel Cells and Electrochemical CO2 Fixation
Pieta, I. S., Kadam, R. G., Pieta, P., Mrdenovic, D., Nowakowski, R., Bakandritsos, A., Tomanec, O., Petr, M., Otyepka, M., Kostecki, R., Khan, M. A. M., Zboril, R., & Gawande, M. B.
Adv. Mater. Interfaces 2021, 2001822
Abstract
S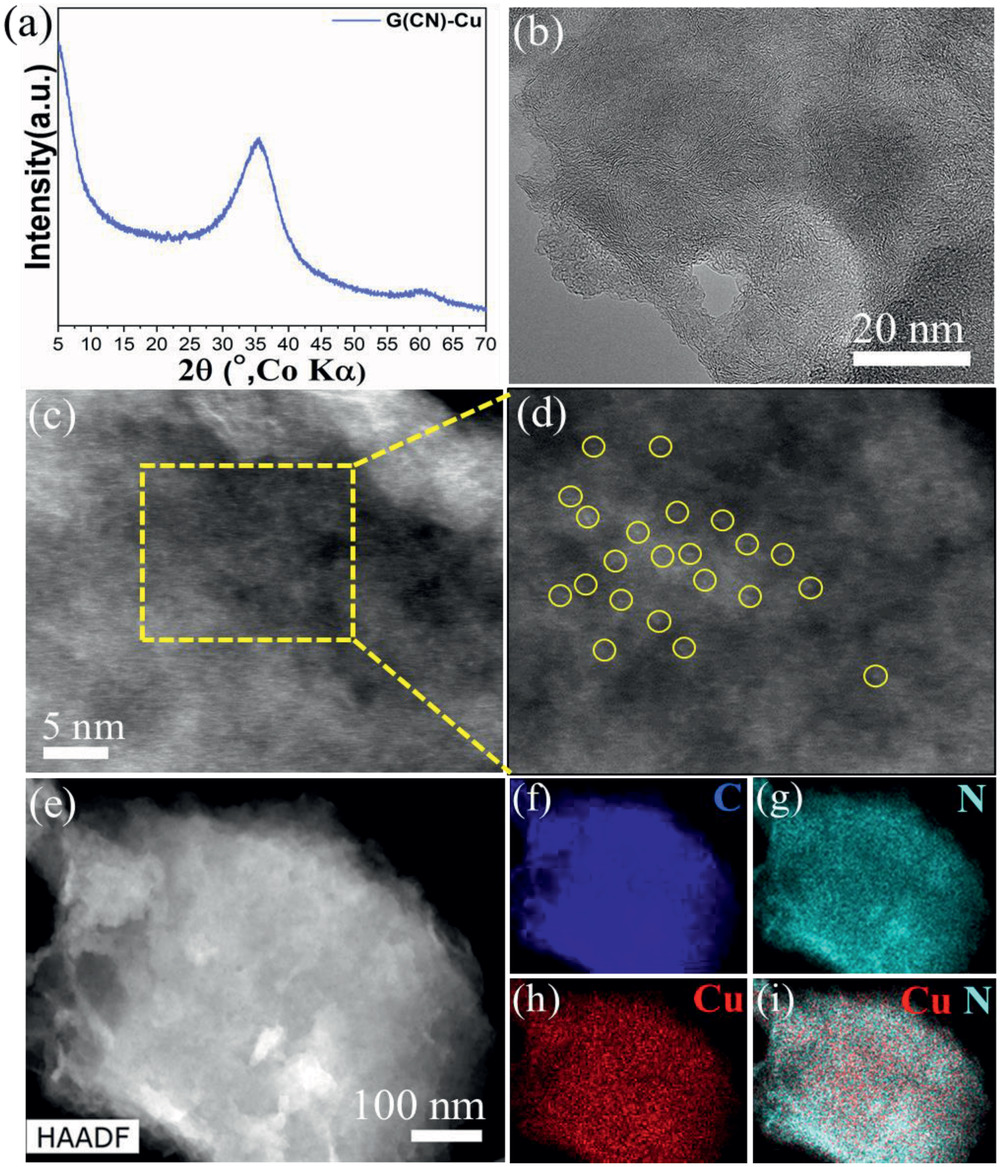 ingle‐atom catalysts (SACs) are highly enviable to exploit the utmost utilization of metallic catalysts; their efficiency by utilizing nearly all atoms to often exhibit high catalytic performances. To architect the isolated single atom on an ideal solid support with strong coordination has remained a crucial trial. Herein, graphene functionalized with nitrile groups (cyanographene) as an ideal support to immobilize isolated copper atoms G(CN)‐Cu with strong coordination is reported. The precisely designed mixed‐valence single atom copper (G(CN)‐Cu) catalysts deliver exceptional conversions for electrochemical methanol oxidation (MOR) and CO2 reduction (CO2RR) targeting a “closed carbon cycle.” An onset of MOR and CO2RR are obtained to be ≈0.4 V and ≈−0.7 versus Ag/AgCl, respectively, with single active sites located in an unsaturated coordination environment, it being the most active Cu sites for both studied reactions. Moreover, G(CN)‐Cu exhibited significantly lower resistivity and higher current density toward MOR and CO2RR than observed for reference catalysts.
ingle‐atom catalysts (SACs) are highly enviable to exploit the utmost utilization of metallic catalysts; their efficiency by utilizing nearly all atoms to often exhibit high catalytic performances. To architect the isolated single atom on an ideal solid support with strong coordination has remained a crucial trial. Herein, graphene functionalized with nitrile groups (cyanographene) as an ideal support to immobilize isolated copper atoms G(CN)‐Cu with strong coordination is reported. The precisely designed mixed‐valence single atom copper (G(CN)‐Cu) catalysts deliver exceptional conversions for electrochemical methanol oxidation (MOR) and CO2 reduction (CO2RR) targeting a “closed carbon cycle.” An onset of MOR and CO2RR are obtained to be ≈0.4 V and ≈−0.7 versus Ag/AgCl, respectively, with single active sites located in an unsaturated coordination environment, it being the most active Cu sites for both studied reactions. Moreover, G(CN)‐Cu exhibited significantly lower resistivity and higher current density toward MOR and CO2RR than observed for reference catalysts.
Figure: Characterization G(CN)‐Cu SACs a) X‐ray powder diffraction (XRD) pattern, b) TEM image, c) representative high‐angle annular dark‐field scanning transmission electron microscopy (HAADF‐STEM) image, d) magnified HAADF‐STEM image showing presence of single atom Cu (highlighted by a yellow circle). e) Another HAADF image of G(CN)‐Cu and the corresponding EDS chemical mapping of G(CN)‐Cu for C (blue) (f), N (cyan) (g), Cu (red) (h), and Cu (red) and N (cyan) together (i).



